News
Breadcrumb
News
Benefits of a flat energy response in a radiation survey meter
A flat energy response means that the survey meter does not give an under- or over-representation of the measured dose at certain energies.

ALARA Rules for Radiation Protection
ALARA is an familiar industry term every medical professional should know and practice. This brief blog post covers the basics of the ALARA principle, and how the RaySafe i3 Real-time dosimeter can help monitor radiation exposure.
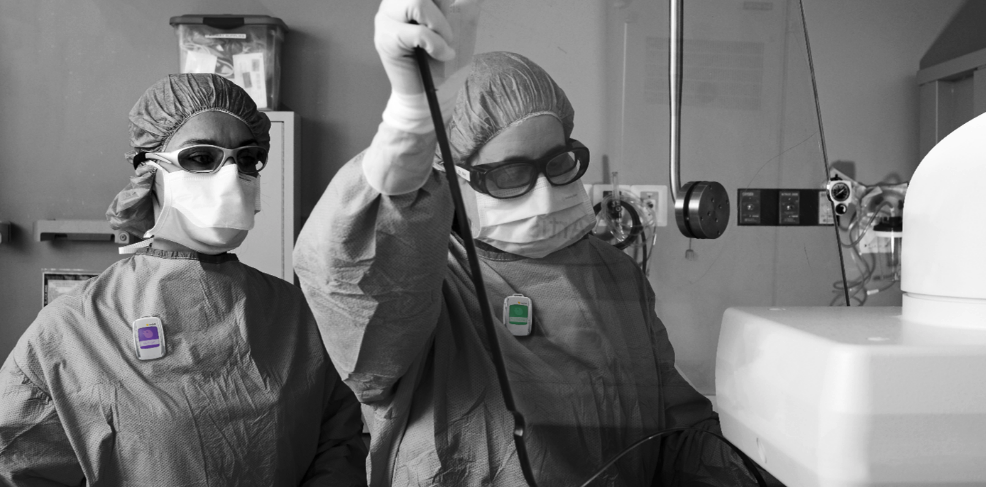
Why Real-time Dosimetry is Important
Personal protection equipment (PPE) has been available for years to help protect medical professionals against ionizing radiation. Most of the time, it consists of heavy protection garments and shields. While these are effective, being able to monitor radiation exposure in real time takes protection to a whole new level.
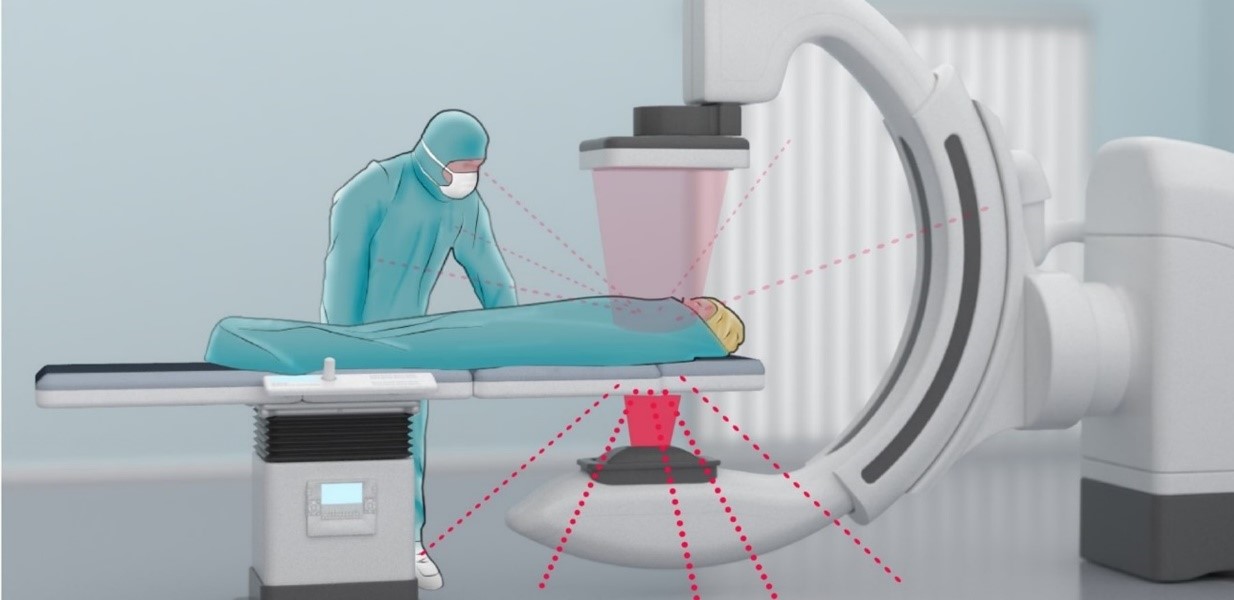
Personal Safety When Working with Ionizing Radiation
Personal radiation safety is paramount within interventional X-ray. Every activity should be planned to minimize unnecessary radiation exposure, as physicians and other staff members are in the room with the patient and run the risk of continued exposure to radiation.

A face-lift, a team, customers' pain, and a vision
We recently launched the RaySafe 452 Survey Meter, a survey meter that is as versatile as our customers, allowing them to measure a variety of applications with a single device.
RaySafe X2 expands with new possibilities - now measure voltage with the new X2 Volt sensor
RaySafe X2 Volt Sensor makes it easier than ever to perform measurements on X-ray machine voltage output test points
Featuring an intuitive interface and automatic waveform capture, the new sensor eliminates the settings and manual adjustments oscilloscopes require
RaySafe 452 Survey Meter is a single, powerful tool that measures radiation for a variety of applications, simplifying data collection
New versatile meter delivers fast, flat, wide energy response times to ensure accurate, trustworthy radiation data collection

Visualizing scattered radiation
As touched upon in the previous post, interventional X-ray professionals have a number of tools and techniques at their disposal, which they can use to minimize their radiation dose.